Empowering health
About Sulfateq B.V.
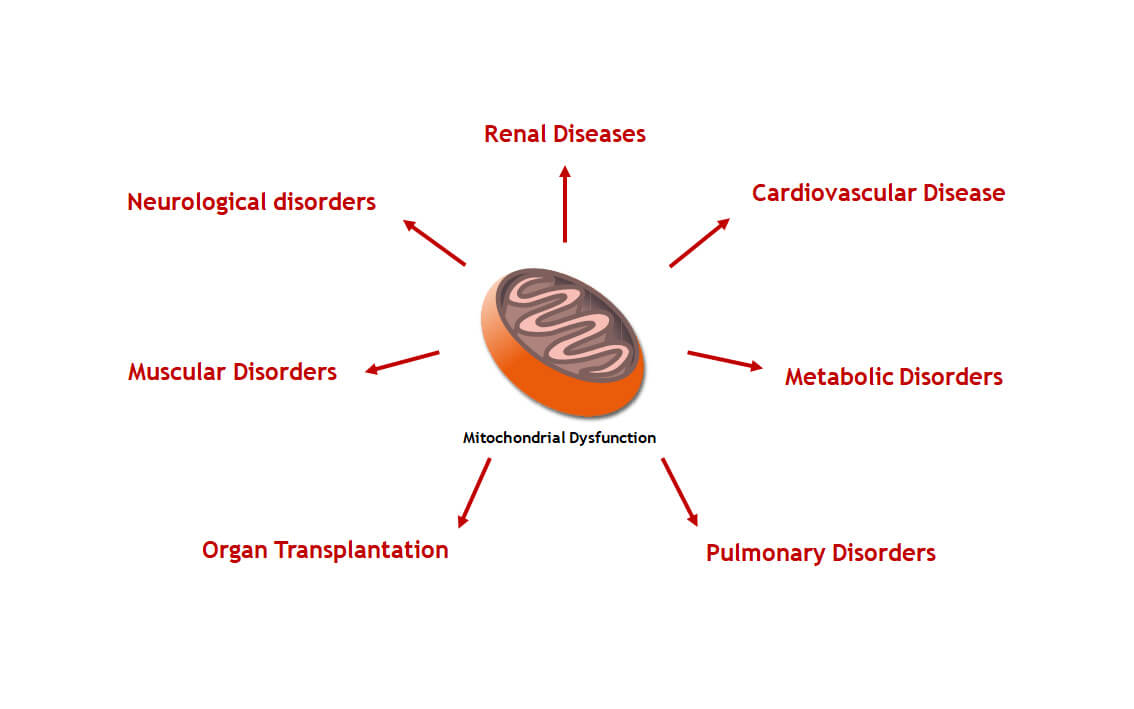
Sulfateq BV is an innovative privately-held biotechnology company that is developing breakthrough compounds for the treatment of a wide range of therapeutic indications related to mitochondrial dysfunction. Our compound library emerged from research on natures natural mechanisms to cope with mitochondrial stress, including hibernation. SUL compounds have an unique mechanism-of-action and the potential to revolutionize the standard-of-care of diseases associated with a declined mitochondrial function.
We strongly believe in the potential of our science to deliver new treatments and improve quality of life for patients with non-communicable diseases involving mitochondrial dysfunction.
We are always open for new innovative partnerships. Please contact us to discuss the opportunities.
GEN Receives Ethical Approval to Launch Phase II Clinical Trial for SUL-238 Targeting Mitochondrial Dysfunction in Patients with Parkinson’s Disease
ANKARA, Türkiye, March 16, 2026 - GEN Pharmaceuticals (GENIL.IS), Türkiye's leading specialty pharmaceutical company, announced today that it has received approval in the Netherlands from the BEBO Foundation for the Assessment of Ethics of Biomedical Research...
Sulfateq B.V.
+31 503137464
info@sulfateqbv.com
Admiraal de Ruyterlaan 5, 9726 GN, Groningen